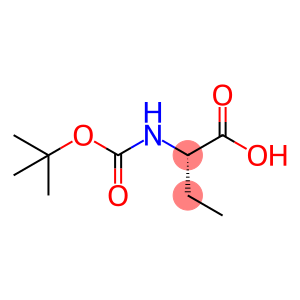
N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid
L-2-(boc-amino)butyric acid
CAS: 34306-42-8
Molecular Formula: C9H17NO4
N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid - Names and Identifiers
| Name | L-2-(boc-amino)butyric acid |
| Synonyms | Boc-Abu-OH Boc-Abu-OH·DCHA RARECHEM EM WB 0016 Boc-L-2-aminobutyric acid L-2-(boc-amino)butyric acid (S)-Boc-2-aminobutyric acid T-BOC-L-2-AMINOBUTANOIC ACID (s)-2-(boc-amino)butyric acid N-T-boc-L-A-aminobutyric acid 2-[(tert-butoxycarbonyl)amino]butanoic acid N-tert-Butoxycarbonyl-L-2-aminobutyric acid (S)-N-T-BUTYLOXYCARBONYL-2-AMINOBUTYRIC ACID N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid (S)-N-TERT-BUTYLOXYCARBONYL-2-AMINOBUTYRIC ACID (2S)-2-[(tert-butoxycarbonyl)amino]butanoic acid N-(tert-butoxycarbonyl)-L-alpha-aminobutyric acid (S)-2-(Boc-amino)butyric acid, Boc-L-2-aminobutyric acid |
| CAS | 34306-42-8 |
| EINECS | 696-164-1 |
| InChI | InChI=1/C9H17NO4/c1-5-6(7(11)12)10-8(13)14-9(2,3)4/h6H,5H2,1-4H3,(H,10,13)(H,11,12)/t6-/m0/s1 |
| InChIKey | PNFVIPIQXAIUAY-LURJTMIESA-N |
N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid - Physico-chemical Properties
| Molecular Formula | C9H17NO4 |
| Molar Mass | 203.24 |
| Density | 1.101±0.06 g/cm3(Predicted) |
| Melting Point | 70-74°C |
| Boling Point | 334.5±25.0 °C(Predicted) |
| Flash Point | 113 °C |
| Water Solubility | Slightly soluble in water. |
| Vapor Presure | 2.42E-05mmHg at 25°C |
| Appearance | Solid |
| Color | White to Almost white |
| BRN | 6801706 |
| pKa | 4.00±0.10(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.46 |
| MDL | MFCD00037267 |
N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S4 - Keep away from living quarters. S7 - Keep container tightly closed. S28 - After contact with skin, wash immediately with plenty of soap-suds. S35 - This material and its container must be disposed of in a safe way. S44 - |
| WGK Germany | 3 |
| HS Code | 29241990 |
| Hazard Class | IRRITANT |
N-(tert-Butoxycarbonyl)-L-a-aminobutyric acid - Reference Information
| Properties | Boc-L-2-aminobutyric acid ((2S)-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid), also known as L-2-(tert-butoxycarbonylamino) butyric acid, its molecular formula is C9H17NO4, molecular weight is 203.24,CAS number is 34306-42-8, melting point 70-74°C, and white powder at room temperature. |
| synthesis | Boc-L-2-aminobutyric acid is generally synthesized from DL-2-aminobutyric acid. the specific synthesis steps are as follows: dissolve 10.3g of DL-2-aminobutyric acid in a mixed solution of 95m L 1M NaOH solution and 65mL of methanol, add 27.5mL of ditert-butyl dicarbonate (I. e. Boc anhydride, 1.2 eq.) into an ice bath, slowly raise to room temperature, and stir for 12 hours. After the reaction is over, methanol is removed by spin evaporation, pH is adjusted to 1-2 for 1M hydrochloric acid, ethyl acetate extraction (50 mLx3), saturated salt water washing (40 mLx2), anhydrous sodium sulfate water removal, and finally spin evaporation to obtain 18.9g colorless solid (93.4%). |
| application example | (1) synthesis of anti-tumor drug idelalisib(2) synthesis of anti-tumor drug edlib (3) synthesis of o-fluoro-o-iminyl benzoic acid intermediate compound |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: 叔丁氧羰基氨基丁酸; Boc-L-2-氨基丁酸; N-Boc-L-2-氨基丁酸; BOC-L-Α-氨基吲哚丁酸; 叔丁氧羰酰基Α氨基丁酸; Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Boc-L-2-aminobutanoic acid Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: BOC-ABU-OH Request for quotation
CAS: 34306-42-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34306-42-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-2-(boc-amino)butyric acid Request for quotation
CAS: 34306-42-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 34306-42-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Boc-L-α-Aminobutyric acid amide Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 叔丁氧羰基氨基丁酸; Boc-L-2-氨基丁酸; N-Boc-L-2-氨基丁酸; BOC-L-Α-氨基吲哚丁酸; 叔丁氧羰酰基Α氨基丁酸; Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Boc-L-2-aminobutanoic acid Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: BOC-ABU-OH Request for quotation
CAS: 34306-42-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 34306-42-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: L-2-(boc-amino)butyric acid Request for quotation
CAS: 34306-42-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 34306-42-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Boc-L-α-Aminobutyric acid amide Visit Supplier Webpage Request for quotationCAS: 34306-42-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




